Achieve Life Sciences Appoints Jeffrey Farrow and Reid Waldman, MD to Board of Directors
Appointments Add Deep Biopharmaceutical Commercial Strategy, Clinical and Finance Expertise as Company Advances Toward P
Read More
We are a late-stage specialty pharmaceutical company dedicated to tackling the global nicotine dependence epidemic by advancing cytisinicline.
If approved, cytisinicline has the potential to become the first new FDA-approved treatment option in 20 years.
Smokers in the US 1
E-Cigs in the US 1
The harms associated with smoking are well established, increasing the risk of COPD, cardiovascular disease, cancer, and other serious conditions.
While over half attempt to quit smoking each year, fewer than 10% succeed.2 The toll is staggering – lost lives and over $600 billion in annual smoking-related healthcare costs.14
A more effective treatment is critical to addressing this widespread public health crisis.
Worldwide Deaths Annually4
Americans Live with Smoking- Related Disease5
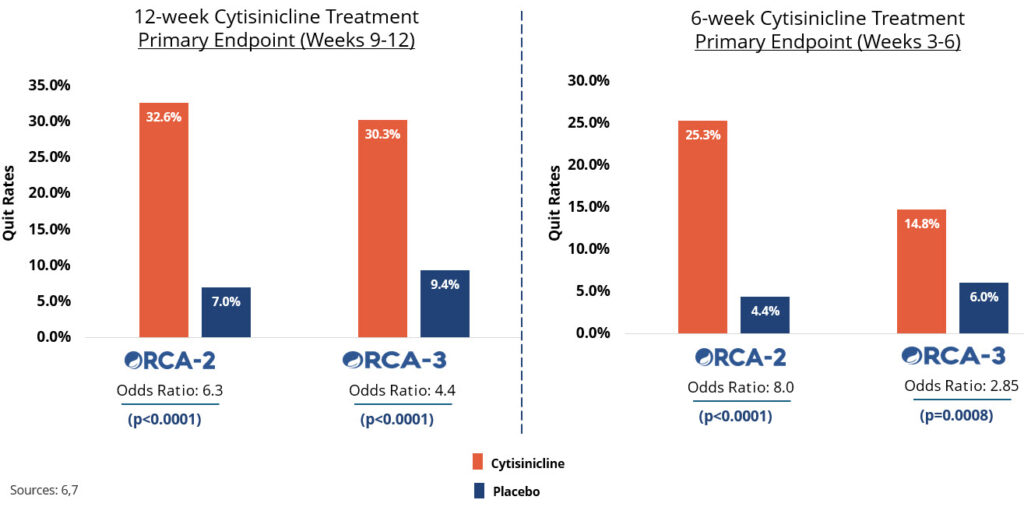
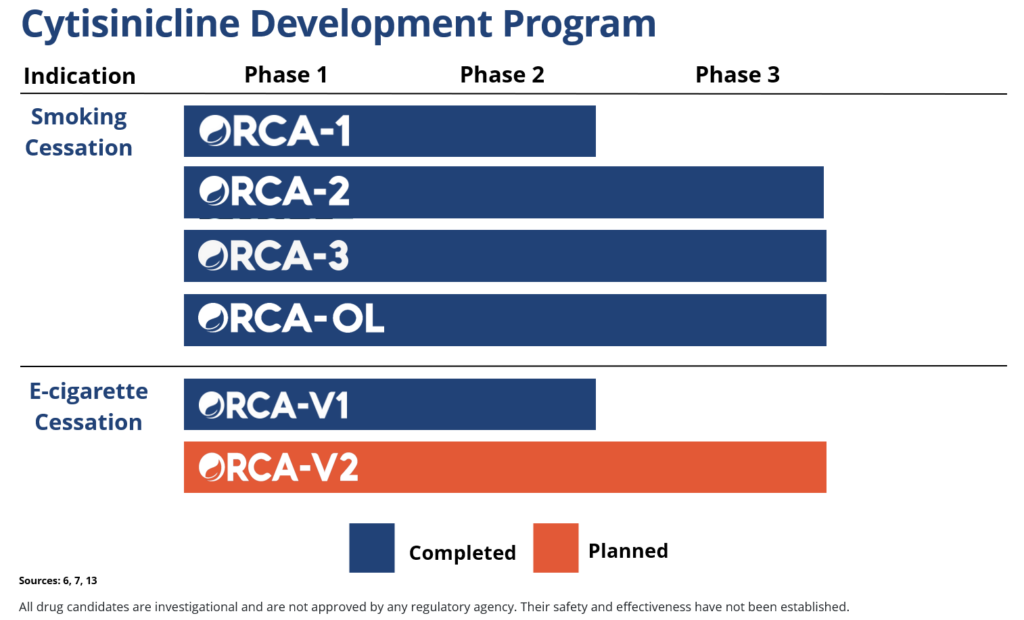
In two recent trials, cytisinicline has been shown to effectively help even highly dependent smokers stop smoking.6,7 Cytisinicline binds to the same receptors as nicotine to reduce both withdrawal symptoms and satisfaction associated with smoking.8 And since it doesn’t bind as strongly to certain receptors, it is believed to have less potential to cause side effects — including nausea and vomiting.


And we’re looking for others, like us, who believe we can bring an end to this fight. Will you join us?